Here’s an interesting scientific development, where a team of scientists “used a process called X-ray crystallography to figure out the exact structure of [hop] acids, humulone molecules, and some of their derivatives, produced from hops in the brewing process.” According to the story in Futurity, they already knew that “beer and its bittering acids, in moderation, have beneficial effects on diabetes, some forms of cancer, inflammation, and perhaps even weight loss.”
The story comes from the journal Angewandte Chemie International Edition and a recent article Absolute Configuration of Beer′s Bitter Compounds. Here’s the opening part of the article.
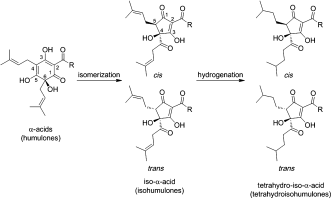
The science and art of making beer, likely the oldest liquid fermented by humans, stretches over millennia. Production typically involves boiling beer wort together with hops, which acts as a natural preservative, but the generated iso-α-acids are known to be prone to decomposition, and consequently, more stable reduced hops extracts, such as the tetrahydro-iso-α-acids, have been developed. These latter compounds are separately produced and frequently added to beer to achieve a consistent level of bitter taste. Scheme 1 gives an overview of the iso-α-acids formed by heat-induced isomerization.
The rest of the story talks about how they isolated the hops in such a way that they might be able to be extracted to use in medicine to take advantage of their healthful properties, that you couldn’t really get just by drinking beer because the amounts in beer were too small to be effective.
The press release from the University of Washington, where the study was conducted, explains the study in layman’s terms.
Humulone molecules are rearranged during the brewing process to contain a ring with five carbon atoms instead of six. At the end of the process two side groups are formed that can be configured in four different ways—both groups can be above the ring or below, or they can be on opposite sides.
Which of the forms the molecule takes determines its “handedness,” Kaminsky says, and that is important for understanding how a particular humulone will react with another substance. If they are paired correctly, they will fit together like a nut and bolt.
If paired incorrectly, they might not fit together at all or it could be like placing a right hand into a left-handed glove. That could produce disastrous results in pharmaceuticals.
Kaminsky cites thalidomide, which has a number of safe uses but was famously used to treat morning sickness in pregnant women in the late 1950s and early 1960s before it was discovered to cause birth defects. Molecule “handedness” in one form of the drug was responsible for the birth defects, while the orientation of molecules in another form did not appear to have the negative effects.
To determine the configuration of humulones formed in the brewing process, coauthors from KinDex Therapeutics, a Seattle pharmaceutical firm that funded the research, recovered acids from the brewing process and purified them.
They converted the humulones to salt crystals and sent them to Kaminsky, who used X-ray crystallography to determine the exact configuration of the molecules.
“Now that we know which hand belongs to which molecule, we can determine which molecule goes to which bitterness taste in beer,” Kaminsky says.
The authors point out that while “excessive beer consumption cannot be recommended to propagate good health, isolated humulones and their derivatives can be prescribed with documented health benefits.”
Some of the compounds have been shown to affect specific illnesses, Kaminsky says, while some with a slight difference in the arrangement of carbon atoms have been ineffective.
The new research sets the stage for finding which of those humulones might be useful in new compounds to be used as medical treatments.
Anybody seeing a new kind of hop pellet? One you take every day with your vitamins, perhaps.


What an idea Jay has! A hop pellet mixed w/malt extract dissolved in a glass of sparkling water for breakfast – beer taste sans alcohol. The possibilities are endless for the mixer/chaser; for those prone to acid reflux, some supplement company could turn the hop pellets into powder & encapsulate 🙂
Check out Metagenics, specifically Kaprex and Insinase…
Clinton
Perhaps some hop flavored hard candy would be the ticket! You never know . . .